|
4/2/2023 0 Comments Atomic number 9
Since sodium has 11 protons, the number of neutrons must be 23 – 11 = 12 neutrons. The mass number of an element tells us the number of protons AND neutrons in an atom (the two particles that have a measurable mass). This tells us that sodium has 11 protons and because it is neutral it has 11 electrons. We know that the atomic number of sodium is 11. /186810031-56a130cd5f9b58b7d0bce8ee.jpg)
the questions silently, again ask them to allotment their account with a 9. The mass number is given at the top left of the elements symbol, for example, sodium has a mass number of 23. as the atomic number increases, the Electronegativity of the elements. This is why elements in the same group of the Periodic Table have similar properties. It is the number of outer electrons that give an element its chemical properties. Fluorine in its elemental state is a highly electronegative element in the periodic. The second energy level is also full, holding eight electrons and one electron remaining in the outer energy level. The first two fill the innermost energy level. The first energy level (the one nearest the nucleus) can hold a maximum of two electrons with the others being able to hold up to a maximum of 8 electrons (only true for the first 20 elements).Ī sodium atom contains 11 electrons orbiting a nucleus in 3 levels, 2 in the lowest, then 8, then 1 All of the electrons in the noble gas neon (atomic number 10) are paired, and all of the orbitals in the n 1 and the n 2 shells are filled. In a neutral atom there are as many electrons as protons moving about nucleus. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10-19 coulombs. These energy levels can only hold a certain number of electrons. Fluorine (atomic number 9) has only one 2p orbital containing an unpaired electron. at end of Appendix B ) Isotope ' Table 1 Table 1 Element ( atomic number ) Col. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. 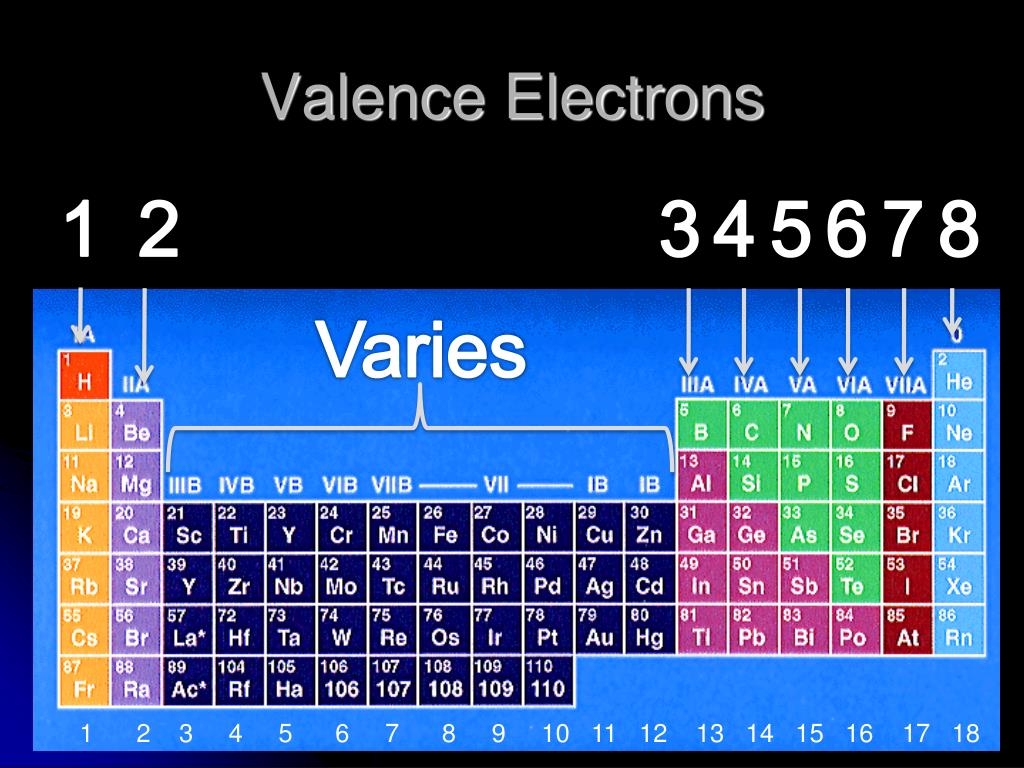
All the electrons are arranged into energy levels. An atom has an atomic number of 9 and a mass number of 19. The electron arrangement of all atoms can be found in the data booklet. Since atoms are neutral, we know then that sodium atoms must also have 11 negative electrons to cancel the charge from 11 positive protons. This is written at the bottom left hand side of the symbol. The atomic number of an element tells you how many protons that the element has.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
